HOME > Program Members > Akihiko Yoshimura

Akihiko Yoshimura

Professor, Department of Microbiology and Immunology, Graduate School of Medicine, Keio University
Akihiko Yoshimura, PhD
yoshimura@a6.keio.jp
http://www.immunoreg.jp/
Theme
Failure of homeostasis of the immune system leads to the onset of various diseases, such as allergic and autoimmune diseases. Immunological tolerance is one of the important mechanisms involved in the maintenance of immunological homeostasis. Foxp3+ regulatory T cells (Treg) and IL-10-expressing regulatory cells (Tr1) play important roles in the tolerance mechanisms to involved in the control of the balance in the activities of the effector T cells. It is believed that fully differentiated effector T cells, such as Th1, Th2 and Th17 cells, are unable to change their effector functions and are resistant to the suppressive actions of Treg. Therefore, a new method(s) to induce re-differentiation of effector cells to a stage similar to naive T cells or a stage sensitive to Treg would contribute to the development of new approaches for the control of various immune diseases. We propose to study the molecular mechanisms that govern the differentiation of T cells in both in vitro and in vivo, with the aim of developing such a novel approach to induce T cell re-differentiation.
Research activities
Various cytokines are involved in the regulation of the immune system and in hematopoiesis. Most cytokines utilize the so-called JAK-STAT pathway, while some activate the Ras-ERK pathway, which plays a more important role than the STAT pathway in the proliferation of hematopoietic cells. Dysregulation of cytokine signaling can cause a variety of diseases, including allergic, inflammatory, and neoplastic diseases. We have identified two important regulator families involved in cytokine signaling: the SOCS proteins and the Spred proteins. Suppressors of the cytokine signaling (SOCS) proteins bind to JAK and to certain receptors, thereby suppressing further signaling events (1). The Spred family proteins interact with Ras and Raf, to suppress ERK activation (2). Studies have shown that the SOCS and Spred proteins are key physiological regulators of immunity, hematopoiesis, and angiogenesis (3, 4). Evidence is also emerging of the the involvement of these proteins in human diseases (5).
- Endo et al. Nature 387: 921-924 (1997).
- Wakioka et al. Nature 412: 647-651 (2001).
- Yasukawa et al.. Nature Immunol. 4: 551-556 (2003).
- Mori H, et al.. Nature Med. 10: 739-743 (2004).
- Brems H, et al. Nature Genetics 39 :1120-6, (2007)
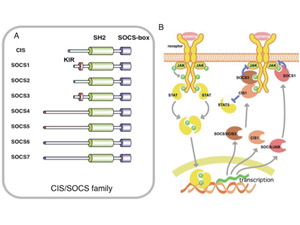
Fig.1 The structure (A) and functions (B) of SOCS proteins
(A) Schematic structure of the CIS/SOCS family of proteins. The SOCS box is conserved in all the proteins of the CIS/SOCS family. SOCS1 and SOCS3 directly inhibit the tyrosine kinase activity of JAK, as they contain a kinase-inhibitory region (KIR) immediately upstream of the central SH2 domain, that is proposed to function as a pseudosubstrate. (B) Mechanism of suppression by CIS, SOCS1 and SOCS3. All of these are induced by cytokine stimulation. CIS1 binds to the STAT5-activating receptors, to suppress further activation of STAT5. SOCS1 binds to the JAKs, and SOCS3 binds to the receptor through the SH2 domain, while both inhibit JAK activity through KIR. These complexes may be degraded by ubiquitination and proteasomal degradation recruited through the SOCS-box.
In-vitro culture system in which the temporal specification of neural development is recapitulated. Using this system, we showed that COUP-TFI/II are required for proper neurogenic-to-gliogenic transition of neural stem cells.
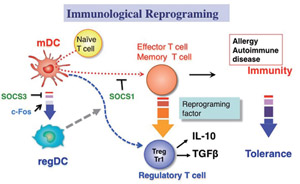
Fig.2 Helper T cells are known as the master regulators of immunity. Dysregulation of the balance between effector and regulatory T cells causes immunological disorders such as allergy and autoimmune diseases. We have discovered the SOCS family proteins, which plays a pivotal role in the maintenance of this balance. In this project, we propose to develop new methods for reprogramming helper as well as memory T cells, that is, conversion of effector and memory T cells to regulatory T cells, as a novel strategy for the regulation of immunological diseases.
Selected Paper
- Brems H, Chmara M, Sahbatou, Denayer, Taniguchi K, Kato R, Somers R, Messiaen L, De Schepper S, Fryns JP, Cools J, Marynen P, Thomas G, Yoshimura A, Legius E. Germline loss-of-function mutations in SPRED1 cause a NF1-like phenotype. Nature Genetics 39 :1120-6, (2007)
- Mori H, Hanada R, Hanada T, Aki D, Mashima R, Nishinakamura H, Torisu T, Chien K.R, Yasukawa H, and Yoshimura A.: SOCS3 deficiency in the brain elevates leptin sensitivity and confers resistance to diet-induced obesity. Nature Med. 10: 739-743 (2004).
- Yasukawa H, Ohishi M, Mori H, Murakami M, Chinen T, Aki D, Hanada T, Takeda K, Akira S, Hoshijima M, Hirano T, Chien K.R, and Yoshimura A.: IL-6 induces an anti-inflammatory response in the absence of SOCS3 in macrophages. Nature Immunol. 4: 551-556 (2003).
- Wakioka T, Sasaki A, Kato R, Shouda T, Matsumoto A, Miyoshi K, Tsuneoka M, Komiya S, Baron R, and Yoshimura A.: Spred, a Sprouty-related suppressor of Ras signaling. Nature 412: 647-651 (2001).
- Endo T.A, Masuhara M, Yokouchi M, Suzuki R, Sakamoto H, Mitsui K, Matsumoto A, Tanimura S, Ohtsubo M, Misawa H, Miyazaki T, Leonor N, Taniguchi T, Fujita T, Kanekura Y, Komiya S and Yoshimura A.: A new protein containing an SH2 domain that inhibits JAK kinases. Nature 387: 921-924 (1997).
Copyright © Keio University. All rights reserved.
