HOME > Program Members > Haruhiko Siomi

Haruhiko Siomi

Professor, Department of Molecular Biology, Graduate School of Medicine, Keio University
Haruhiko Siomi, PhD
awa403@sc.itc.keio.ac.jp
http://web.sc.itc.keio.ac.jp/dmb/sindex.html
Theme
In a variety of eukaryotic organisms, sequence information in long RNA transcripts is converted into small non-coding RNAs such as microRNAs (miRNAs), small interfering RNAs (siRNAs), and piwi-interacting RNAs (piRNAs). These small RNAs trigger various forms of sequence-specific gene silencing by guiding effector complexes or RISCs to target RNAs via base-pairing. This process is collectively referred to as RNA silencing. Mutations in RNA silencing-pathway genes often disrupt stem cell maintenance.
Argonaute proteins are a defining component of RISC complexes and fall into two classes; the AGO subfamily and the PIWI subfamily. Drosophila has three distinct PIWI proteins (AGO3, Aub, Piwi), that are primarily expressed in the germline. Germline stem cells (GSCs) can self-renew and generate differentiated progeny. It is known that GSC maintenance and division require piwi expression in the somatic cells that form the stem cell niche.
Our objective is to use a combination of biochemical approaches and genetic experiments to understand in detail how cellular and molecular interactions between GSCs and the stem cell niche regulate self-renewal and differentiation of GSCs.
Research activities
It was nearly a decade ago when we found that a Drosophila homolog of fragile X mental retardation protein (FMR1) is associated with components of RNA silencing, including the fly Argonaute, AGO2. Argonaute proteins are a defining component of RISC complexes. Since then, we have produced specific monoclonal antibodies directed against distinct Argonaute proteins in Drosophila, mice and humans, in order to exploit biochemical approaches to understand the molecular pathways in RNA silencing. Using these antibodies, we have identified new components and elucidated their functions in RNA silencing via the miRNA, endo-siRNA and piRNA pathways. It is becoming clear that RNA silencing plays very important roles in the self-renewal and differentiation of stem cells. With 'next generation' sequencing technologies, we expect our anti-Argonaute antibodies to become very powerful tools to help define the fundamental principles of stem cell regulation.
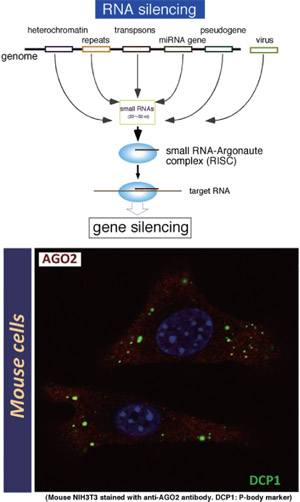
Fig.1
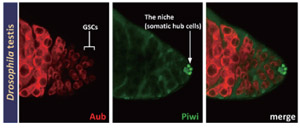
Fig.2
Selected Paper
- Azuma-Mukai, A., Oguri, H., Kin, T., Qian, ZR., Asai, K., Siomi, H., and Siomi, MC. 2008. Characterization of endogenous human Argonautes and their miRNA partners in RNA silencing. Proc. Natl. Acad. Sci. USA. 105:7964-7969.
- Kawamura, Y., Saito, K., Kin, T., Ono, Y., Asai, K., Sunohara, T., Okada, NT., Siomi MC. & Siomi, H. 2008. Drosophila endogenous small RNAs bind to Argonaute2 in somatic cells. Nature 453: 793-797.
- Gunawardane, LS., Saito, K., Nishida, KM., Miyoshi, K., Kawamura, Y., Nagami, T., Siomi, H., and Siomi, MC. 2007. A Slicer-mediated mechanism for rasiRNA 5'end formation in Drosophila. Science 315: 1587-1590.
- Saito, K., Nishida, KM., Mori, T., Kawamura, Y., Miyoshi, K., Nagami, T., Siomi, H., and Siomi, MC. 2006. Specific association of Piwi with rasiRNAs derived from retrotransposon and heterochromatic regions in the Drosophila genome. Genes & Development 20: 2214-2222.
- Miyoshi, K., Tsukumo, H., Nagami, T., Siomi, H., and Siomi, MC. 2005 Slicer function of Drosophila Argonautes and its involvement in RISC formation. Genes & Development 19: 2837-2848.
Copyright © Keio University. All rights reserved.
