HOME > Program Members > Yasunori Okada

Yasunori Okada

Professor, Department of Pathology, Graduate School of Medicine, Keio University
Yasunori Okada, MD, PhD
okada@sc.itc.keio.ac.jp
http://keio-okada-lab.jp/
Theme
Regulation of tissue repair, inflammation and tumorigenesis and cancer progression by modulation of tissue microenvironment
Cellular functions in vivo are strictly regulated by the tissue microenvironment, which is composed of the extracellular matrix (ECM) and ECM-associated bioactive factors, such as growth factors, cytokines and chemokines. These tissue microenvironmental factors function through cell-cell junctions, cell-cell interactions, cell-ECM interactions, and the actions of autocrine and paracrine (Figure 1). Fibrosis through metabolism of the growth factors, cytokines and chemokines and niche formation are important topics in inflammation and tissue repair. In addition, growth factor metabolism by tumor-associated fibroblasts, and tissue reactions such as angiogenesis contribute to tumorigenesis and cancer progression. We shall study the mechanisms of regulation of proteinases responsible for tissue microenvironmental modulation, with the aim of developing novel methods to control tissue repair, inflammation, and tumorigenesis and cancer progression by focusing on cancer stem cells (Figure 2).
Research activities
We studied the biochemical characteristics of MMPs and ADAMs and analyzed their functional roles in pathological conditions. Our data indicate that MMP-2 plays a key role in cardiac rupture after acute myocardial infarction and suggested the possibility of control of myocardial remodeling by modulation of the MMP-2 activity. We also obtained data suggesting that MMP-7 and ADAM12 contribute to chondrocyte repair in osteoarthritic cartilage. ADAM28 was selectively overexpressed in carcinoma cells in the human breast and non-small cell lung carcinomas. We showed that ADAM28 enhances the release of insulin-like growth factor-I (IGF-I) from the IGF-I/IGF binding protein-3 (IGFBP-3) complex by selective digestion of IGFBP-3 and promotes carcinoma cell proliferation. In addition, ADAM28 enhances P-selectin glycoprotein ligand-1(PSGL-1)/P-selectin-mediated cell rolling adhesion to endothelial cells by interaction with PSGL-1, suggesting a role of this protease in cancer cell metastasis.
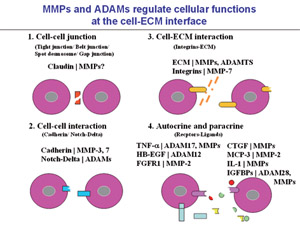
Fig.1 Regulation of cellular functions by modulation of cell-cell junctions, cell-cell interactions, cell-ECM interactions, and autocrine and paracrine factors by MMPs and ADAMs.MMPs and/orADAMs can digest the molecules involved in these interactions and modulate the cellular functions.
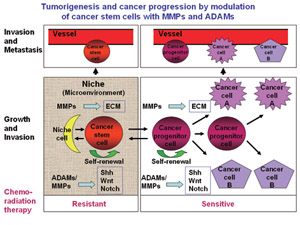
Fig.2 The stemness of cancer stem/progenitor cells is maintained in their tissue microenvironment (niche),probably through interaction of cancer stem/progenitor cells with niche cells and/or ECM.
MMPs and ADAMs may modulate their functions by cleaving and/or interacting with thet issue microenvironmental factors.
Selected Paper
- Okada Y.: Proteinases and matrix degradation. In Kelley's Textbook of Rheumatology. Ed. by Harris E. D., Jr., Budd R. C., Ruddy S., Genovese M.C., Firestein G. S. and Sargent J. S. 8th edition, Elsevier Saunders. Philadelphia. p115-134, 2009.
- Okada A., Mochizuki S., Yatabe T., Kimura T., Shiomi T., Fujita Y., Matsumoto H., Sehara-Fujisawa A., Iwamoto Y. and Okada Y.: ADAM12 (Meltrin-α) is involved in chondrocyte proliferation via cleavage of insulin-like growth factor-binding protein-5 in osteoarthritic cartilage. Arthritis Rheum. 58:778-789, 2008.
- Shimoda M., Hashimoto G., Mochizuki S., Ikeda E., Nagai N., Ishida S. and Okada Y.: Binding of ADAM28 to P-selectin glycoprotein ligand-1 enhances P-selectin-mediated leukocyte adhesion to endothelial cells. J. Biol. Chem. 282: 25864-25874, 2007.
- Mitsui Y., Mochizuki S., Kodama T., Shimoda M., Ohtsuka T., Shiomi T., Chijiiwa M., Ikeda T., Kitajima M. and Okada Y.: ADAM28 is overexpressed in human breast carcinomas: Implications for carcinoma cell proliferation through degradation of insulin-like growth factor binding protein-3. Cancer Res. 66:9913-9920, 2006.
- Matsumura S., Iwanaga S., Mochizuki S., Okamoto H., Ogawa S. and Okada Y.: Targeted deletion or pharmacological inhibition of MMP-2 prevents cardiac rupture after myocardial infarction in mice. J. Clin. Invest. 115:599-609, 2005.
Copyright © Keio University. All rights reserved.
