HOME > Program Members > Yasuhide Morikawa

Yasuhide Morikawa

Professor, Department of Surgery, Graduate School of Medicine, Keio University
Yasuhide Morikawa, MD, PhD
ymorikaw@sc.itc.keio.ac.jp
http://web.sc.itc.keio.ac.jp/surgery/ps/index.html
Theme
Hirschsprung's disease is a developmental disorder of the enteric nervous system in the gut and its main symptoms are constipation and abdominal distension in newborns. The pathogenesis of this disease is thought to involve migration arrest of neural crest-derived cells resulting in congenital absence of normal enteric nerves, known as ganglion cells, in the distal segment of the gut. Neural crest stem cells (NCSCs) are known as the origin of ganglion cells and also play key roles in the repair and maintenance of ganglion cells. Until date, the only treatment available for this condition is surgery, which requires removal of the non-functional part of the bowel. We have also performed small bowel transplantation for intractable cases. However, even after operation, the children often suffer from long-term problems. In the present study, the authors are involved in examining the possibility of transplantation of NCSCs into the aganglionic bowel as a novel therapeutic strategy for Hirschsprung's disease. We shall first attempt to isolate NCSCs from the murine embryonic gut. Then, our plan is to investigate the efficacy of NCSC transplantation using a mouse model of Hirschsprung's disease and human gut samples.
Research activities
The present studies were carried out using experimental mice and clinical samples.
[Isolation of neural crest (stem) cells from the gut]
First, we attempted to isolate NCSCs from the murine gut. In order to obtain neural crest-derived cells efficiently, transgenic mice with GFP-labeling of the neural crest-derived cells were used. GFP-positive (neural crest-derived) cells were successfully isolated from the fetal gut and spheres were formed.
[Differentiation assay]
- Mice
The spheres obtained exhibited the expression of markers for smooth muscle (SMA), neurons (TuJ1) and glial cells (GFAP), consistent with the trilineage differentiation pattern of neural crest stem cells (NCSCs) (Fig. 1-.a). - Humans
Gut-derived cells were isolated and sphere formation was observed. The spheres exhibited the expression of markers for smooth muscle (SMA), neurons (PGP9.5 and TuJ1) and glial cells (GFAP) (Fig. 1-.b).
[Co-culture of murine neural crest sphere with fetal colon]
The obtained spheres were transplanted into the murine embryonic colon in a co-culture fashion. It was found that the GFP-positive cells colonized in the recipient colon and migrated downwards. A nerve-network-like migration showing GFP fluorescence was observed. These GFP-positive cells also exhibited positivity for PGP9.5. These results demonstrate that the transplanted neural crest spheres differentiated into enteric nerves.
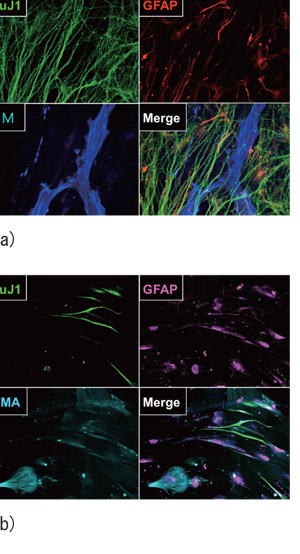
Fig.1 Mouse and human neural crest-derived spheres showing trilineage differentiation.
(a)The mouse neural crest-derived spheres differentiated into neurons (TuJ1), glial cells (GFAP), and smooth muscle (SMA).
(b)The human gut-derived spheres differentiated into neurons (TuJ1),glial cells (GFAP), and smooth muscle (SMA).
Selected Paper
- Watanabe T, Hoshino K, Tanabe M, Morikawa Y, Kawachi S, Wakabayashi G, Shimazu M, Kitajima M.:Correlation of motility and neuronal integrity with a focus on the grade of intestinal allograft rejection. Am J Transplant. 2008 Mar;8(3):529-36. Erratum in: Am J Transplant. 2008 Jun;8(6):1355. Am J Transplant. 2008 Jun;8(6):1356.
- Komori K, Fuchimoto Y, Morikawa Y, Obara H, Kawachi S, Tanabe M, Hoshino K,Shimazu M, Matsuzaki Y, Kitajima M.:The role of graft and host accommodation in a hamster-to-rat cardiac transplantation model. Transplantation. 2008 Jan 15;85(1):112-7.
- Oguma J, Ozawa S, Morikawa Y, Furukawa T, Kitagawa Y, Ueda M, Kitajima M.:Knot-tying force during suturing and wound healing in the gastrointestinal tract. J Surg Res. 2007 Jun 1;140(1):129-34.
- Yamayoshi Y, Watanabe T, Tanabe M, Hoshino K, Matsumoto K, Morikawa Y,Shimadzu M, Kitajima M, Tanigawara Y.:Novel application of ProteinChip technology exploring acute rejection markers of rat small bowel transplantation. Transplantation. 2006 Aug 15;82(3):320-6.
- Shimojima N, Nakaki T, Morikawa Y, Hoshino K, Ozaki H, Hori M, Kitajima M.:Interstitial cells of Cajal in dysmotility in intestinal ischemia and reperfusion injury in rats. J Surg Res. 2006 Oct;135(2):255-61.Epub 2006 Jul 26.
Copyright © Keio University. All rights reserved.
